| Names | |
|---|---|
| Other names PVOH; Poly(Ethenol), Ethenol, homopolymer; PVA; Polyviol; Vinol; Alvyl; Alcotex; Covol; Gelvatol; Lemol; Mowiol; Mowiflex, Alcotex, Elvanol, Gelvatol, Lemol, Mowiol, Nelfilcon A, Polyviol und Rhodoviol | |
| Identifiers | |
| |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | |
| E number | E1203 (additional chemicals) |
| KEGG | |
| RTECS number |
|
| Properties | |
| (C2H4O)x | |
| Density | 1.19-1.31 g/cm3 |
| Melting point | 200 °C (392 °F; 473 K) |
| log P | 0.26 [1] |
Refractive index (nD) | 1.477 @ 632 nm[2] |
| Hazards | |
| Safety data sheet | External MSDS |
| NFPA 704 (fire diamond) | |
| Flash point | 79.44 °C (174.99 °F; 352.59 K) |
| Lethal dose or concentration (LD, LC): | |
| 14,700 mg/kg (Mouse) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| verify (what is ?) | |
| Infobox references | |
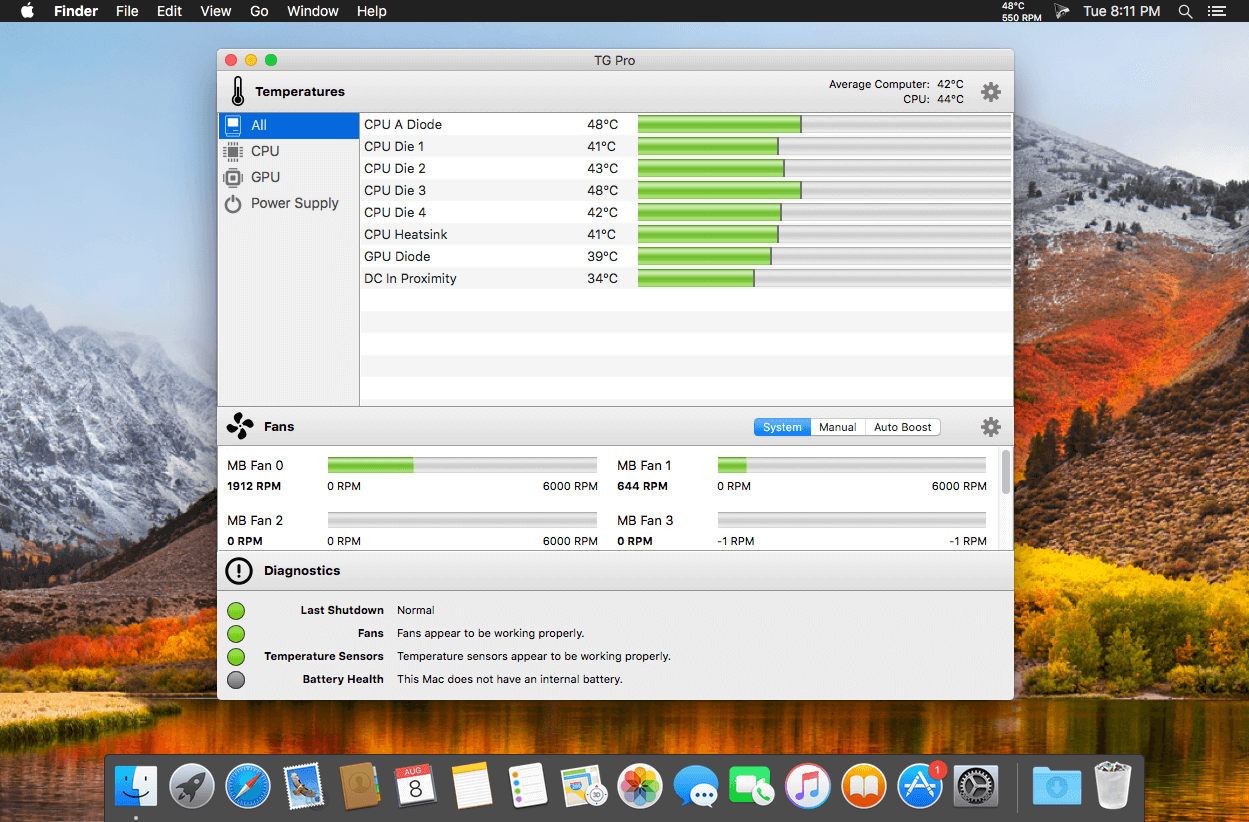
By March 1991, NEC claimed that it had sold 750,000 TurboGrafx-16 consoles in the United States and 500,000 CD-ROM units worldwide. In an effort to relaunch the system in the North American market, in mid-1992 NEC and Hudson Soft transferred management of the system in North America to a new joint venture called Turbo Technologies Inc. And released the TurboDuo, an all-in-one unit that. Temperature Gauge Pro is a well-designed application for the macOS platform created from the ground up to enable you to monitor the temperature of all heat sensors in your Mac. Moreover, Temperature Gauge Pro can be used to keep track and control the fan speeds, run diagnostics on. Install Advanced Uninstaller PRO; 2. Start Advanced Uninstaller PRO. Take your time to admire Advanced Uninstaller PRO's design and wealth of features available. Advanced Uninstaller PRO is a very useful Windows tool. Simply hdr 3 2 6. Click on the General Tools category 4. Press the Uninstall Programs tool 5. All the programs installed on the PC will appear. TG Pro (Temperature Gauge Pro) is a must-have utility for every Mac user with five awesome features: Check up on your Mac by viewing the current and maximum recorded temperatures for every sensor. Choose between Celsius or Fahrenheit. Tune the fan speeds so that your Mac can run cooler with the manual fan control.
Poly(vinyl alcohol) (PVOH, PVA, or PVAl) is a water-solublesynthetic polymer. It has the idealized formula [CH2CH(OH)]n. It is used in papermaking, textile warp sizing, as a thickener and emulsion stabilizer in PVAc adhesive formulations and a variety of coatings. It is colourless (white) and odorless. It is commonly supplied as beads or as solutions in water.[3][4]
Uses[edit]
PVA is used in a variety of medical applications because of its biocompatibility, low tendency for protein adhesion, and low toxicity. Specific uses include cartilage replacements, contact lenses, and eye drops.[5] Polyvinyl alcohol is used as an aid in suspension polymerizations. Its largest application in China is its use as a protective colloid to make polyvinyl acetate dispersions. In Japan its major use is the production of Vinylon fiber.[6] This fiber is also manufactured in North Korea for self-sufficiency reasons, because no oil is required to produce it. Another application is photographic film.[7]
Nowadays PVA-based polymers are being used widely in additive manufacturing. For example, 3D printed oral dosage forms demonstrate great potential in the pharmaceutical industry. It is possible to create drug-loaded tablets with modified drug-releas characteristics where PVA is used as a binder substance.[8]
Medically, it may also used as the embolic agent in a Uterine Fibroid Embolectomy (UFE) [9]
Polyvinyl acetals[edit]
Polyvinyl acetals are prepared by treating PVA with aldehydes. Butyraldehyde and formaldehyde afford polyvinyl butyral (PVB) and polyvinyl formal (PVF), respectively. Preparation of polyvinyl butyral is the largest use for polyvinyl alcohol in the U.S. and Western Europe.
Tg Pro 2 392 Stroker Kit
Preparation[edit]
Unlike most vinyl polymers, PVA is not prepared by polymerization of the corresponding monomer as the monomer, vinyl alcohol, is thermodynamically unstable with respect to its tautomeracetaldehyde. Instead, PVA is prepared by hydrolysis of polyvinyl acetate,[3] or sometimes other vinyl ester-derived polymers with formate or chloroacetate groups instead of acetate. The conversion of the polyvinyl esters is usually conducted by base-catalysed transesterification with ethanol:
- [CH2CH(OAc)]n + C2H5OH → [CH2CH(OH)]n + C2H5OAc
The properties of the polymer are affected by the degree of transesterification.
Worldwide consumption of polyvinyl alcohol was over one million metric tons in 2006.[6] Large producers include Kuraray (Japan, Europe, and US) and Sekisui Specialty Chemicals (US), while mainland China has installed a number of very large production facilities in the past decade and currently accounts for 45% of world capacity.
Structure and properties[edit]
PVA is an atactic material that exhibits crystallinity. Memory cleaner 1 2 – ram cleaner near me. In terms of microstructure, it is composed mainly of 1,3-diol linkages [-CH2-CH(OH)-CH2-CH(OH)-] but a few percent of 1,2-diols [-CH2-CH(OH)-CH(OH)-CH2-] occur, depending on the conditions for the polymerization of the vinyl ester precursor.[3]
Tg Pro 2 392 Engine
Polyvinyl alcohol has excellent film forming, emulsifying and adhesive properties. It is also resistant to oil, grease and solvents. It has high tensile strength and flexibility, as well as high oxygen and aroma barrier properties. However, these properties are dependent on humidity: water absorbed at higher humidity levels acts as a plasticiser, which reduces the polymer's tensile strength, but increases its elongation and tear strength.
Safety and environmental considerations[edit]
PVA is widely used, thus its toxicity and biodegradation are of interest. Solutions containing up to 5% PVA are nontoxic to fish.[3] It biodegrades slowly.[10]
See also[edit]
References[edit]
Tg Pro 2 392 Hemi
- ^'Poly(vinyl alcohol)_msds'.
- ^Schnepf MJ, Mayer M, Kuttner C, Tebbe M, Wolf D, Dulle M, et al. (July 2017). 'Nanorattles with tailored electric field enhancement'. Nanoscale. 9 (27): 9376–9385. doi:10.1039/C7NR02952G. PMID28656183.
- ^ abcdHallensleben ML (2000). 'Polyvinyl Compounds, Others'. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_743.
- ^Tang X, Alavi S (2011). 'Recent Advances in Starch, Polyvinyl Alcohol Based Polymer Blends, Nanocomposites and Their Biodegradability'. Carbohydrate Polymers. 85: 7–16. doi:10.1016/j.carbpol.2011.01.030.
- ^Baker MI, Walsh SP, Schwartz Z, Boyan BD (July 2012). 'A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications'. Journal of Biomedical Materials Research. Part B, Applied Biomaterials. 100 (5): 1451–7. doi:10.1002/jbm.b.32694. PMID22514196.
- ^ abSRI Consulting CEH Report Polyvinyl Alcohol, published March 2007, abstract retrieved July 30, 2008.
- ^Lampman S (2003). Characterization and Failure Analysis of Plastics. ASM International. p. 29.
- ^Xu X, Zhao J, Wang M, Wang L, Yang J (August 2019). '3D Printed Polyvinyl Alcohol Tablets with Multiple Release Profiles'. Scientific Reports. 9 (1): 12487. doi:10.1038/s41598-019-48921-8. PMC6713737. PMID31462744.
- ^Siskin GP (16 November 2018). Cho KJ (ed.). 'Uterine Fibroid (Leiomyoma) Embolization and Imaging'. WebMD LLC.
- ^Kawai F, Hu X (August 2009). 'Biochemistry of microbial polyvinyl alcohol degradation'. Applied Microbiology and Biotechnology. 84 (2): 227–37. doi:10.1007/s00253-009-2113-6. PMID19590867. S2CID25068302.
External links[edit]
Tg Pro 2 392 Horsepower
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Polyvinyl_alcohol&oldid=983845659'
